
- For printing sterilisation labels
- Secure labelling of sterile goods
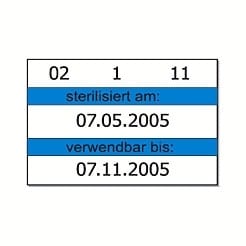
- Indication of batch number, sterilisation & expiry date
- Includes 1 label roll of 750 labels
- Easy and quick handling
MELAdoc Label Printer for Sterilisation Packages
The MELAdoc label printer simplifies labelling, documentation of the release of packaged sterilised instruments and textiles and enables batch traceability: By indicating the sterilisation date, batch number, the name of the person releasing the instruments for use and the steriliser used, the sterilised instruments can be easily assigned to the patient and the sterilisation batch. Use the MELAdoc - label printer in conjunction with documentation sheets (e.g. MELAdoc documentation sheets).
MELAdoc labels are double self-adhesive. After using the instruments, the MELAdoc label can simply be removed from the packaging and stuck in the patient file.
The flawless packages containing the sterilised items are marked after sterilisation by applying a label. This fulfils the prerequisites for proper "release" by the assistant entrusted with reprocessing. The risky labelling of film packaging with ballpoint pens (push-through) or permanent markers (microperforation due to loosening of the plastic) is avoided.
With the MELAdoc label printer you can label sterile goods quickly and safely. The labels contain information about the sterilisation date, expiry date, operator and batch number. A roll with 750 labels is included in the delivery.
Product Details
- MELAdoc label printer for sterilisation packaging from Melag
- For printing sterilisation labels
- Labelling and documentation of the release of sterilised instruments & textiles
- Enables batch tracking
- Double self-adhesive labels
- Fast and secure labelling of sterile goods
- Label details: sterilisation date, expiry date, operator, batch number
- Includes one roll with 750 labels
Traceability
In the event of an allegation of secondary infection, all information about the correct sterilisation process can be assigned to the instruments used via the MELAdoc labels in the patient file. Reference to the person responsible for reprocessing and release and the device used (steriliser number) can be established, information on sterilisation date and batch number enables the assignment to the sterilisation protocols and thus reliable proof: the instruments used were sterilised properly.
Product Identification
Manufacturer: Melag
MPN: 01095
Art. No.: 432148
Share your experiences with this product with other customers. Please click on the stars to submit your rating.










.jpg)




